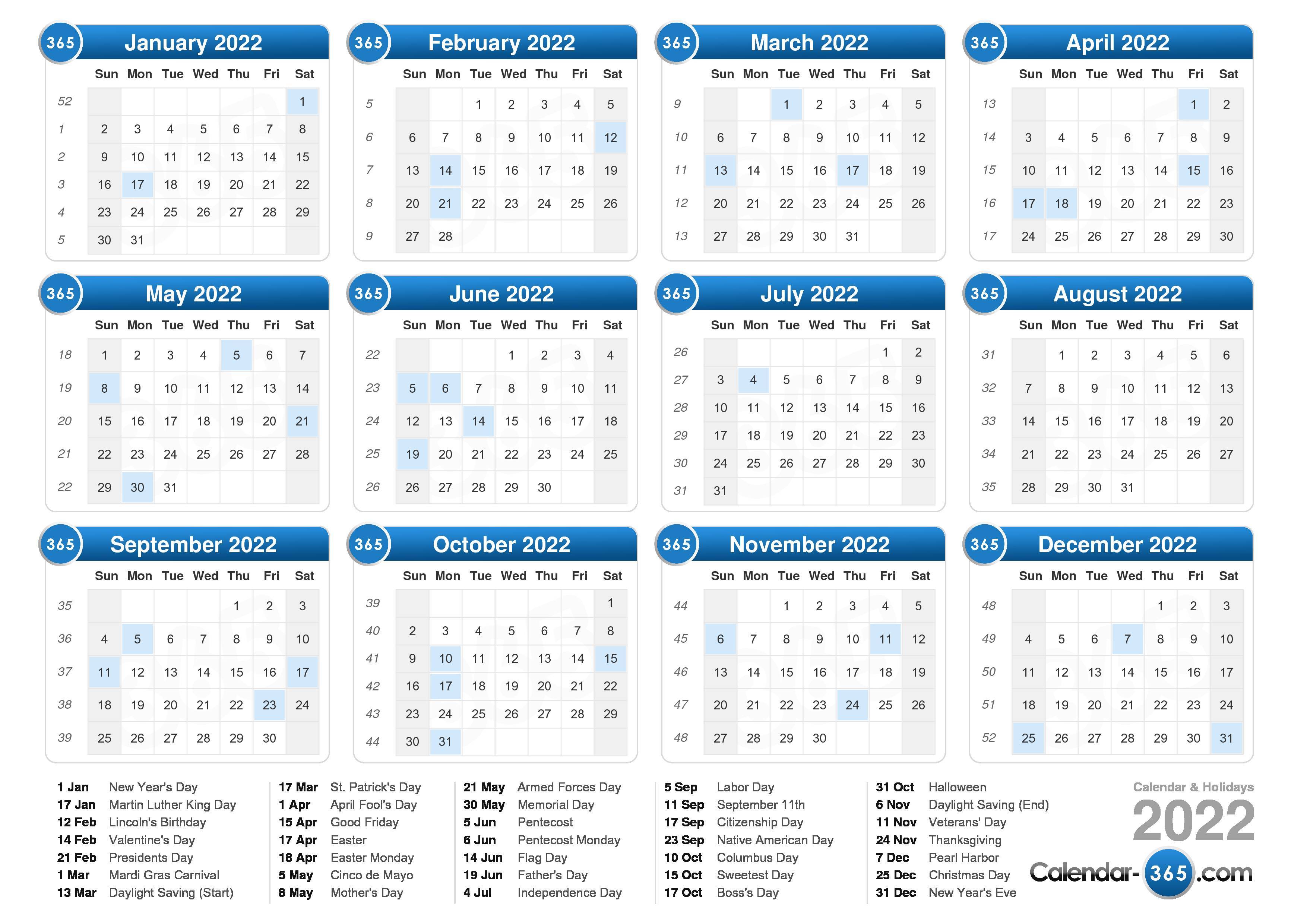
Valoctocogene Roxaparvovec Gene Therapy for Hemophilia A
5
$ 11.00
In stock
(693)
Product Description

FDA Approves First Gene Therapy for Severe Hemophilia A - Southern Iowa Mental Health Center
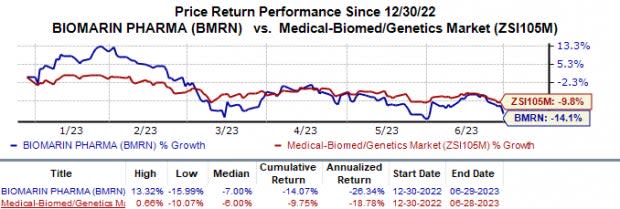
BioMarin (BMRN) Gets FDA Nod for Hemophilia A Gene Therapy
Hemostatic Response is Maintained for up to 5 Years Following Treatment with Valoctocogene Roxaparvovec, an AAV5-hFVIII-SQ Gene Therapy for Severe Hemophilia A - ISTH Congress Abstracts

Gene therapy for hemophilia: a review on clinical benefit, limitations, and remaining issues - ScienceDirect
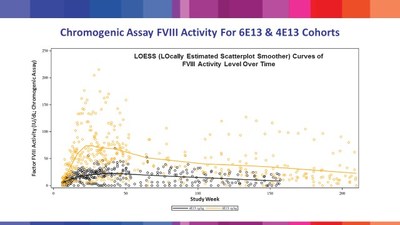
BioMarin Provides Additional Data from Recent 4 Year Update of Ongoing Phase 1/2 Study of Valoctocogene Roxaparvovec Gene Therapy for Severe Hemophilia A in Late-Breaking Oral Presentation at World Federation of Hemophilia
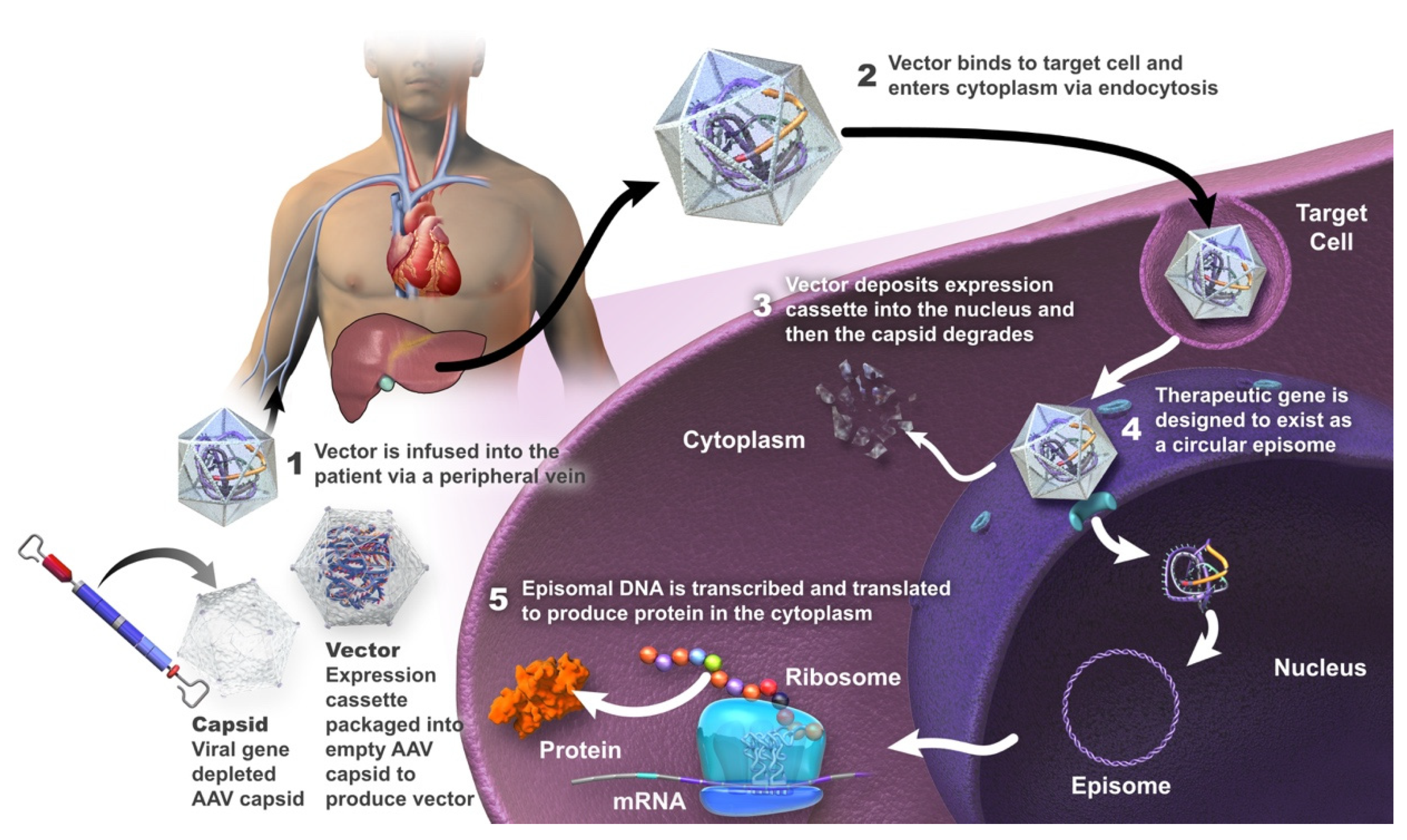
IJMS, Free Full-Text

FDA approves valoctocogene roxaparvovec gene therapy for adults with severe hemophilia A

A Molecular Revolution in the Treatment of Hemophilia - ScienceDirect
BioMarin Begins Mid Stage Study on Gene Therapy Candidate

Early Phase Clinical Immunogenicity of Valoctocogene Roxaparvovec, an AAV5-Mediated Gene Therapy for Hemophilia A: Molecular Therapy
Benefits of Valoctocogene Roxaparvovec Persist in Hemophilia A - Consumer Health News

Matching‐adjusted indirect comparison of bleeding outcomes in severe haemophilia A: Comparing valoctocogene roxaparvovec gene therapy, emicizumab prophylaxis, and FVIII replacement prophylaxis - Astermark - 2023 - Haemophilia - Wiley Online Library

Valoctocogene Roxaparvovec for Severe Hemophilia A, Pipeline